Acid-Base Titration Worksheet
A Grade 10 science worksheet covering acid-base titration principles, calculations, and laboratory procedures.
Includes
Standards
Topics
Acid-Base Titration Worksheet
Name:
Date:
Score:
Read each question carefully and answer to the best of your ability. Show all calculations where necessary.
1. What is the primary purpose of an acid-base titration?
To determine the pH of a solution.
To find the concentration of an unknown acid or base.
To produce a salt and water.
To measure the temperature change during a reaction.
2. Which piece of laboratory equipment is used to accurately dispense the titrant?
Volumetric flask
Beaker
Burette
Pipette
3. The point at which the moles of acid equal the moles of base in a titration is called the .
4. An indicator changes at or near the equivalence point.
5. A student performs a titration of 25.0 mL of an unknown concentration of HCl with 0.100 M NaOH. If 18.5 mL of NaOH is required to reach the equivalence point, what is the concentration of the HCl solution? Show your work.
6. A strong acid-strong base titration will always have an equivalence point at pH 7.
True
False
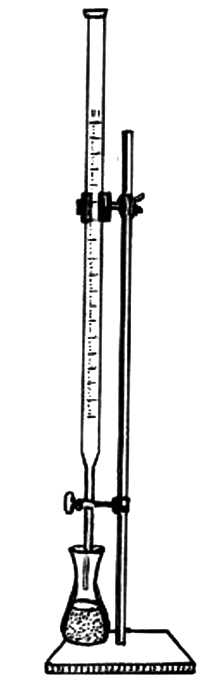
7. Label the following components of a titration setup: