Titration Fundamentals Worksheet
This worksheet covers the fundamental principles and practical aspects of acid-base titrations, including calculations and indicator selection, suitable for Grade 11 Chemistry students.
Includes
Standards
Titration Fundamentals
Name:
Date:
Score:
Read each question carefully and provide your answers in the space provided. Show all your work for calculations.
1. What is the primary purpose of a titration?
To determine the pH of a solution.
To find the concentration of an unknown solution.
To prepare a standard solution.
To measure the volume of a liquid.
2. Which piece of glassware is used to deliver a precise, variable volume of liquid in a titration?
Volumetric flask
Beaker
Burette
Pipette
1. The point at which the indicator changes color during a titration is called the .
2. In an acid-base titration, a reaction occurs between the acid and the base.
3. A solution of known concentration, used to determine the concentration of an unknown solution, is called a solution.
1. Describe the difference between the equivalence point and the endpoint in a titration.
2. Explain why an indicator is necessary for most acid-base titrations.
Observe the image below, depicting a burette, a key piece of equipment in a titration experiment. Then answer the questions that follow.
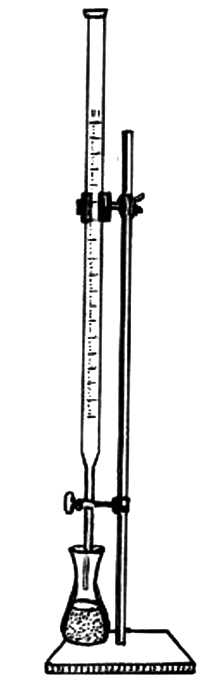
1. What is the primary function of the stopcock on a burette?
2. Why is it important to read the meniscus at eye level when using a burette?
In a titration, 25.0 mL of an unknown concentration of HCl is titrated with 0.150 M NaOH. If 32.5 mL of NaOH is required to reach the equivalence point, what is the concentration of the HCl solution? (Show all steps of your calculation).