Grade 12 Redox Equations Worksheet
This worksheet covers fundamental concepts and applications of redox reactions, including identifying oxidation states, balancing equations, and understanding electrochemical cells.
Includes
Standards
Topics
Grade 12 Redox Equations Worksheet
Name:
Date:
Score:
Read each question carefully and provide your best answer. Show all your work for calculations and balancing equations.
1. In a redox reaction, the species that loses electrons is said to be:
Reduced
Oxidized
Neutralized
Hydrolyzed
2. Which of the following statements is true about the oxidizing agent in a redox reaction?
It loses electrons.
It is oxidized.
It gains electrons.
Its oxidation number decreases.
3. The process of oxidation involves a(n) in oxidation number.
4. A substance that causes another substance to be oxidized is called a(n) agent.
5. Determine the oxidation number of sulfur in the following compounds:
a) H₂SO₄:
b) SO₂:
6. Balance the following redox reaction in acidic solution:
Cr₂O₇²⁻(aq) + Fe²⁺(aq) → Cr³⁺(aq) + Fe³⁺(aq)
7. In the reaction Zn + Cu²⁺ → Zn²⁺ + Cu, zinc is the reducing agent.
True
False
8. Observe the image below depicting a redox reaction. Identify the species being oxidized and the species being reduced.
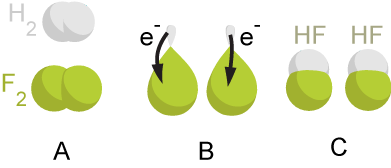
Oxidized Species:
Reduced Species:
9. Explain the difference between an electrochemical cell and an electrolytic cell, including how redox reactions occur in each.