Stereochemistry of Alkene Additions
Grade 12 Chemistry worksheet on the stereochemical outcomes of various alkene addition reactions, including syn and anti additions, and their implications for product formation.
Includes
Standards
Topics
Stereochemistry of Alkene Additions
Name:
Date:
Score:
Read each question carefully and provide detailed answers. For reaction mechanisms, show all relevant stereochemistry using wedges and dashes where appropriate.
1. Which of the following reactions typically proceeds with syn addition?
Halogenation (Br2 addition)
Hydroboration-oxidation
Acid-catalyzed hydration
Oxymercuration-demercuration
2. The addition of Br2 to trans-2-butene yields primarily:
A meso compound
A racemic mixture of enantiomers
A single enantiomer
No reaction occurs
3. The addition of HBr to 1-butene follows Markovnikov's rule, where the hydrogen adds to the carbon with hydrogens.
4. Anti addition implies that the two new substituents are added to faces of the double bond.
5. Explain why hydroboration-oxidation of an alkene results in an anti-Markovnikov syn addition of water. Include the key intermediate that dictates the stereochemical outcome.
6. Draw the complete reaction mechanism, including all stereochemistry (using wedges and dashes), for the addition of Br2 to cis-2-butene. Predict the major stereoisomeric product(s).
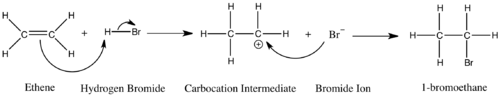
7. The formation of a bromonium ion intermediate during halogenation of an alkene is responsible for the observed anti addition.
True
False