Ionic Compound Formulas Worksheet
Grade 9 Chemistry worksheet focusing on writing and understanding ionic compound formulas.
Includes
Standards
Topics
Ionic Compound Formulas
Name:
Date:
Score:
Read each question carefully and provide the best answer. Remember to consider the charges of ions when writing formulas.
Observe the illustration below showing the formation of an ionic bond.
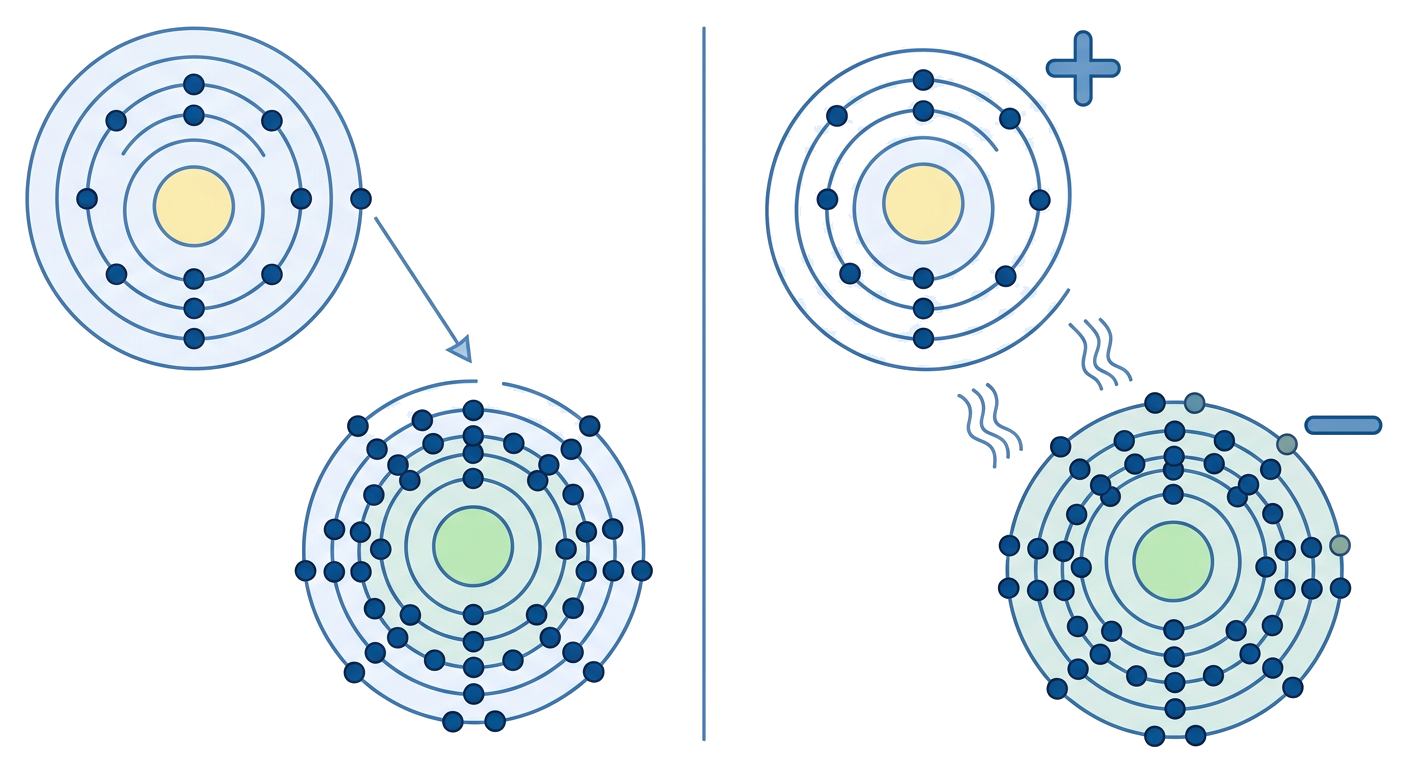
1. Which of the following best describes an ionic bond?
Sharing of electrons between two nonmetals.
Transfer of electrons from a metal to a nonmetal.
Attraction between two positively charged ions.
Formation of a covalent network solid.
2. What is the correct formula for the ionic compound formed between Magnesium (Mg) and Oxygen (O)?
MgO2
Mg2O
MgO
Mg(OH)2
3. An ionic compound is formed between a metal and a .
4. When writing the formula for an ionic compound, the total positive charge must the total negative charge.
5. The positive ion in an ionic compound is called a , and the negative ion is called an .
6. Write the chemical formula for the ionic compound formed between Sodium (Na) and Chlorine (Cl). Explain your reasoning.
7. Determine the ionic formula for Aluminum (Al) and Sulfate (SO4). Show your work.
8. Ionic compounds typically have high melting points.
True
False
9. Polyatomic ions are single atoms with a charge.
True
False
Match the metal ion with its correct charge.
10. Potassium
a. 2+
11. Iron (II)
b. 1+
12. Aluminum
c. 3+
Related Worksheets
Single Replacement Reactions
Explore the principles of single replacement reactions in chemistry with this comprehensive worksheet for Grade 9 students.
History of Chemistry Timeline
Explore key discoveries and scientists that shaped the field of chemistry over time.
Hydrocarbons Worksheet
A Grade 9 science worksheet covering the basics of hydrocarbons, including their definition, types, and properties.
Nuclear Changes: Grade 9 Science
Explore the fundamentals of nuclear changes, including radioactivity, nuclear fission, and nuclear fusion, with this comprehensive Grade 9 science worksheet.
Molecular Compound Naming Worksheet
Practice naming molecular compounds using prefixes and suffixes.
Empirical and Molecular Formula Worksheet
Grade 9 Chemistry worksheet focusing on understanding and calculating empirical and molecular formulas.
VSEPR Theory Worksheet
Explore VSEPR theory with this worksheet, covering molecular geometries, electron domains, and bond angles for various molecules.
Acid Base Nomenclature Worksheet
A Grade 9 science worksheet on acid and base nomenclature, covering common acids, bases, and pH concepts.